Password Reset
Forgot your password? Enter the email address you used to create your account to initiate a password reset.
Forgot your password? Enter the email address you used to create your account to initiate a password reset.
8 Minutes
Intravenous fluids play a critical role in the management of acutely ill patients as a bolus infusion for resuscitation, a continuous maintenance fluid, and as a vehicle to administer medications. It is now well established that acutely ill patients are at risk for developing hospital-acquired hyponatremia due to numerous stimuli for arginine vasopressin secretions (AVP), and that isotonic solutions should be used and hypotonic solutions should be avoided in order to prevent hospital-acquired hyponatremia.
1 One area of controversy is whether isotonic intravenous solutions should be 0.9% saline (normal saline) versus a balanced electrolyte solution (lactated Ringer’s or Plasma-Lyte).2 There has been a growing concern that 0.9% saline has a supraphysiologic chloride concentration and may result in untoward complications, such as hyperchloremic metabolic acidosis, renal vasoconstriction, delayed micturition, hyperkalemia, and an increased incidence of acute kidney injury and need for renal-replacement therapy. No well-conducted clinical trials have been able to demonstrate the superiority of a balanced solution over 0.9% saline in clinical practice until two recent studies published in the New England Journal of Medicine.
Two large trials recently published in the New England Journal of Medicine sought to address whether balanced solutions are superior to 0.9% saline in clinic practice; the data did suggest that there was a small benefit to balanced resuscitation solutions in comparison to normal saline.3,4 The Saline Against Lactated Ringer’s or Plasma-Lyte in the Emergency Department (SALT-ED) and Isotonic Solutions and Major Adverse Renal Events (SMART) trials involved almost 30,000 adult patients at one large tertiary care center in the United States. These were parallel pragmatic clinic trials where there was a waiver of informed consent, fluid allocation was unblinded to either normal saline or a balanced solution based on alternating calendar months, and the volume of the resuscitation solution and choice of balanced solution (either lactated Ringer’s or Plasma-Lyte) was left to the discretion of the treating physician. In the SMART study, patients were admitted to one of five different ICUs. Both studies demonstrate an absolute decrease of about 1 percent for a composite of major adverse kidney events at 30 days, defined as death, new renal replacement therapy, or a persistent two-fold increase in serum creatinine. Neither study was able to demonstrate a statistically significant benefit to any one factor independently. The benefits of a balanced solution were seen primarily in critically ill patients with sepsis and those with preceding acute kidney injury and previous renal replacement therapy. Two groups of patients that fared better with 0.9% saline were patients admitted to the cardiac ICU or with traumatic brain injury.
Based on the findings of these two studies, many editorial commentaries and news outlets have recommended that balanced solutions be used in favor of normal saline.5 Given that millions of hospitalized patients require fluid resuscitation, even a modest benefit, with a need to treat 100 patients to prevent one major adverse renal event, could potentially translate into a benefit to tens of thousands of patients yearly. By extension, practitioners may choose to use balanced solutions over normal saline for not only resuscitation fluids, but as maintenance fluids as well, as there is no obvious theoretical advantage to using normal saline over a balanced solution.
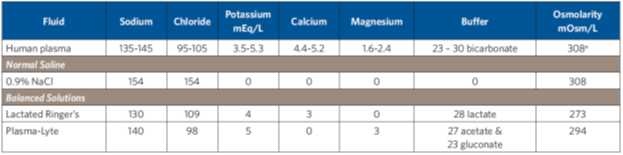
a The osmolality for plasma is 275-295 mOsm/kg
Balanced solutions differ from normal saline in a few important ways. Foremost is that they have variable amounts of a buffering agent, such as lactate, acetate, or gluconate. Balanced solutions do not have bicarbonate, as it is not stable in polyvinyl chloride bags. Balanced solutions also have variable amounts of potassium, calcium, and magnesium, and have a lower sodium concentration and osmolarity in comparison to normal saline (See Table 1 on Page 3). 0.9% saline (Na 154 mmol/L) has the same sodium concentration as the aqueous phase of plasma, whereas Plasma-Lyte (Na 140 mmol/L) and lactated Ringer’s (Na 130 mmol/L) are slightly hypo- tonic in relationship to plasma. Plasma is 93% aqueous and 7% anhydrous, consisting of proteins and lipids.
British physician Sydney Ringer developed a 0.75% saline as a frog tissue preservation solution in the 1870s.6 The fluid was slightly hypotonic, as frogs have a lower plasma sodium concentration (105 mmol/L) in comparison to humans. Dutch physiological chemist Hartog Jakob Hamburger developed 0.9% saline (154 mmol/L) in the 1890s, which has a similar sodium concentration to the aqueous phase of plasma in humans and mammals (151 mmol/L). 0.9% saline came into clinical practice in the 1910s. It was not until the 1930s that a version of Ringer’s solution came into clinical practice, when American pediatrician Alexis Hartmann modified Ringer’s solution by adding lactate in order to treat acidosis in children with diarrheal dehydration. Therefore, lactated Ringer’s solution (sodium 130 mmol/L) is a hyponatremic solution in relationship to the aqueous phase of plasma and is known to contribute to hyponatremia.7 Plasma-Lyte has a combined sodium, potassium, and magnesium concentration of 148 mmol/L, which is very similar to the sodium concentration of normal saline (154 mmol/L).
The SALT-ED and SMART studies did not attempt to address which balanced solution is superior.8 The study design and analysis pooled the data for lactated Ringer’s and Plasma-Lyte together and did not evaluate the benefits and complications as separate variables. In both studies, there was significantly more hyponatremia associated with balanced solutions in comparison to normal saline, which is likely due to the use of lactated Ringer’s. In the SALT-ED, 95 percent of patients received lactated Ringer’s, and the serum sodium fell (p <0.001). In the SMART study, on the other hand, only 44 percent received lactated Ringer’s, with a significant variation of fluid choice in the five ICUs. 94 percent admitted to the medical ICU received lactated Ringer’s, whereas a minority in the other four ICUs (surgical 38 percent, cardiac 24 percent, trauma 18 percent, and neurosurgical 2 percent) received lactated Ringer’s. The incidence of hyponatremia in SMART was higher for balanced solutions (38.1 percent) compared to normal saline (35.5 percent) (p=0.002). The hyponatremic effect of balanced solution was seen even though the median volume of resuscitation fluids administered in both trials was relatively small at only 1000 mL.
It is encouraging that a simple change in fluid therapy from normal saline to a balanced solution could potentially translate into a clinical benefit with decreased acidosis and adverse renal events, but this might come at cost if lactated Ringer’s is the solution that is used. Most practitioners are probably more familiar with lactated Ringer’s than Plasma-Lyte and might assume that the fluids are equivalent. Lactated Ringer’s is a hyponatremic solution and an increase in its use could increase the incidence of hospital-acquired hyponatremia. Hyponatremia is an independent predictor of hospital mortality and is associated with increased hospital costs, length of hospital stay, and rates of readmission. Hyponatremia can be difficult to treat, so preventive measures should be taken by avoiding intravenous fluids with free water, like lactated Ringer’s. Further studies are underway to better evaluate the potential benefit and complications associated with different balanced solutions. Until the results of those studies are available, Plasma-Lyte would appear to be the preferred balanced resuscitation solution due to its higher sodium concentration.