Password Reset
Forgot your password? Enter the email address you used to create your account to initiate a password reset.
Forgot your password? Enter the email address you used to create your account to initiate a password reset.
14 Minutes
Case Report
We describe the case of a bright student who presented to the UPMC Children’s Hospital of Pittsburgh Pediatric Sleep Evaluation Center with a common sleep complaint — an inability to wake up on time for school. A subsequent comprehensive workup of the patient led to the revelation of an unusual and poorly understood sleep problem.
Summary of Case
AB is a 16-year-old boy without any significant past medical history who was referred to the Pediatric Sleep Evaluation Center by his pediatrician and neurologist because of the onset of difficulty waking up for school and the exhibition of floppiness upon awakening. AB was barely responsive upon awakening and also was unable to walk. He did not remember any efforts to arouse him until he “woke up” on the way to school. The patient reported eight hours of sleep on school nights with > 13 hours of catch-up sleep with a late wake-up time on weekends.
These symptoms coincided with the onset of puberty and were accompanied by a prolongation of sleep duration that was interfering with his ability to complete schoolwork. He did not have daytime sleepiness; his Epworth sleepiness scale (ESS) score of 5 (< 10 considered normal) was in the normal range. He reported occasional sleep paralysis but no cataplexy, hypnagogic hallucinations, REM-related (rapid eye movement) phenomena, or restless legs. He did report consistent snoring. AB did not have a history of viral illnesses, head trauma, or new medications. His overall physical examination was unremarkable.
AB had a normal neurology evaluation and a normal video EEG. Polysomnography did not reveal obstructive sleep apnea. His initial actigraphy showed an average daily sleep time of eight hours in the first week and nine to 10 hours in the second week, with delayed wake time on weekends by more than three hours.
As the initial picture was consistent with delayed sleep phase syndrome with inadequate total sleep time on weekdays, he was referred to one of the sleep psychologists in the Pediatric Sleep Evaluation Center.
AB worked diligently with the sleep psychologist to improve his sleep schedule. However, his symptoms, specifically the inability to wake up in the morning, continued to worsen with a
concomitant increase in sleep duration. His father was now taking two or more hours to get him out of bed. The father had a video of AB sitting on the stairs and sleeping while his father attempted to take him to the car to drive him to school. Three hours after awakening, he “woke up” and did not feel the need to sleep anymore during the day. He did not report daytime sleepiness, felt refreshed after sleep, and his ESS had not changed. His total sleep time had increased further to 10.5 hours on weekdays and 12 hours on weekends confirmed by a sleep diary and actigraphy testing (Figures 1 and 2). He still had delayed wake times on weekends by two hours, which was somewhat of an improvement from the previous time of three hours.
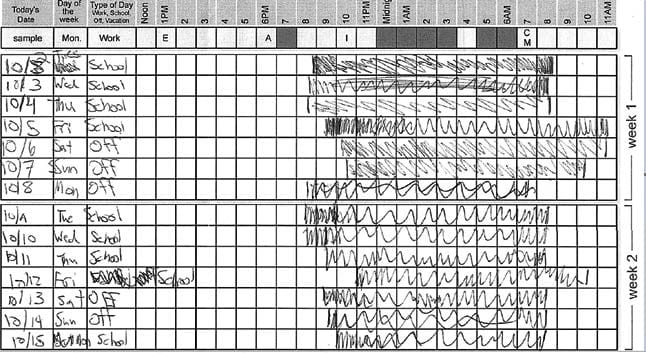
Figure 1. Sleep Diary.
Polysomnography with multiple sleep latency testing (MSLT) to evaluate sleepiness showed an overnight sleep time of 433 minutes and sleep efficiency of 86 percent with an arousal index of 15 per hour; apnea-hypopnea index (AHI) 0.8 per hour; and oxygen saturation above 90 percent for 100 percent of the night. The sleep study was terminated at 6 a.m. per protocol. Upon awakening from the overnight study, the patient reported that he could not move his legs and was not alert for some time after the lights came on. During the MSLT, AB only slept on the first nap and had a mean sleep latency (MSL) of 19 minutes without any REM sleep.
Discussion
In summary, AB had new-onset excessive sleep inertia or “sleep drunkenness” (with confirmed prolonged sleep duration) in conjunction with normal subjective sleepiness during the day and normal nap latency without REM periods. This new-onset constellation of symptoms with progression of sleep needs was felt to be most consistent with idiopathic hypersomnia (IH), with the exceptions that AB found sleep refreshing and did not feel sleepy during the day. We discuss our reasoning in the context of physiological sleep requirements and central disorders of hypersomnolence.
Physiologic Sleep Requirements
Adequate duration and quality of sleep are important for health and well-being. Short sleep duration is associated with a wide range of negative health outcomes including accidents, poor work performance, obesity, and increased risk of self-harm or suicidal thoughts. The length of nocturnal sleep depends on multiple factors including intrinsic/genetic determinants and volitional control.1 While sleep restriction is generally regarded as detrimental to health, optimal sleep duration is not clearly understood. There are several competing sets of recommendations for the optimal duration of sleep, and a commonly cited reference is from the National Sleep Foundation (NSF), which also is endorsed by the American Academy of Pediatrics (https://www.sleepfoundation.org/press-release/national-sleep-foundation-recommends-new-sleep-times). These consensus guidelines developed by sleep experts recommend eight to 10 hours of sleep for teenagers, with less than seven hours and more than 11 hours “not recommended.” Of note, sleep requirements steadily decrease from 14 to 17 hours during infancy to seven to nine hours in adulthood.
Hypersomnia
Excessive sleepiness or hypersomnolence is a common sleep complaint that refers to the inability to stay awake during major wake periods of the day resulting in an excessive need for sleep, unscheduled periods of drowsiness, or sleep present despite normal quality and timing of nocturnal sleep.2 It is important not to confuse symptoms of fatigue/ tiredness with those of true sleepiness. Chronic fatigue syndrome has persistent fatigue and a subjective report of tiredness that does not resolve with sleep or rest.
The third edition of the International Classification of Sleep Disorders (ICSD) classifies central disorders of hypersomnolence into eight subtypes2:
• Narcolepsy type 1 and 2 (NT1 and NT2)
• Idiopathic hypersomnia (IH)
• Kleine-Levin syndrome
• Hypersomnia due to medications
• Hypersomnia due to a medical disorder
• Hypersomnia in relation to a psychiatric disorder
• Insufficient sleep syndrome and normal variant — long sleeper (physiological long sleeper)
In addition to the presence of episodes of rapid onset uncontrollable daytime sleepiness, NT1 is characterized by cataplexy and/or hypocretin deficiency and MSL ≤ 8 minutes and two or more sleep onset REM periods (SOREMP). NT2 has polysomnographic and MSLT features of NT1 without cataplexy or low levels of hypocretin.2 Klein-Levin syndrome is defined as episodes of recurrent, time-limited hypersomnia (two days to five weeks) associated with cognitive or perceptual dysfunction, disinhibition, or disordered eating with a return to normal baseline between events.3 Insufficient sleep syndrome is completely reversed by normalizing sleep time. Physiological long sleepers have long sleep periods from early childhood, feel fully rested with sleep, and do not have any sleepiness if they are able to satisfy their sleep requirements. Hypersomnia associated with medical or psychiatric disorders or medication occurs in conjunction with relevant triggers.
Idiopathic Hypersomnia
Compared to narcolepsy, which was first described in 1877, the identification of IH is very recent and, in fact, the term itself was coined as late as 1976.4 Since then, there have been several variations in the definition of the disease, including the newly released ICSD 3, which revised the diagnostic criteria and simplified the definition of IH as supported by underlying data. The current ICSD 3 IH criteria are: 1) periods of daytime lapses into sleep; 2) absence of cataplexy; 3) fewer than two SOREMPs during MSLT; and 4) symptoms not explained by other causes including insufficient sleep. Additionally, one or both of the following should be present: 1) MSL of eight minutes or less during the MSLT, and/or 2) 11 hours or more of sleep per day (either in a 24-hour sleep study or in a week of at-home monitoring). Sleep drunkenness and long unrefreshing naps are supportive features.2
IH is an uncommon disorder that affects mostly young subjects with an uncertain prevalence with estimates of 0.002% to 0.02% of the population. In comparison, the prevalence of excessive daytime sleepiness in the general population ranges from 4% to 30%. The average age of onset of IH is reported from 16.6 to 21.2 years, and the disorder is generally stable and persistent though spontaneous remission has been reported.
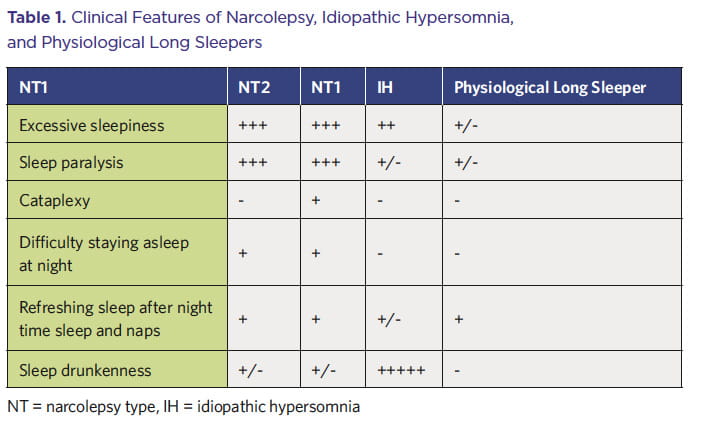
Debilitating daytime sleepiness is a feature of IH though it is reported to be less irresistible than with narcolepsy (See Table 1 on Page 4). Naps tend to be longer and unrefreshing in 48% to 78% of patients with IH, and self-reported sleep duration is 10 hours or more in 30% of patients. Sleep efficiency on polysomnography is usually high (90% to 94%).
Sleep inertia refers to the transitional state between sleep and wake on awakening, marked by impaired performance, reduced vigilance, and a desire to return to sleep. Some sleep inertia is a normal phenomenon but can have dangerous ramifications, e.g., in health care workers5 or military personnel who are woken abruptly in the night and required to make cognitively taxing decisions.6
Pronounced sleep inertia or sleep drunkenness is defined as a prolonged difficulty in awakening with repeated returns to sleep, irritability, automatic behaviors, and confusion. It is a core supportive feature of IH with symptoms lasting up to four hours. It can be more problematic than daytime sleepiness with some patients requiring the assistance of another person to be able to wake up. Sleep drunkenness is present in 1% of the general population compared to 36% to 66% of patients with IH.7
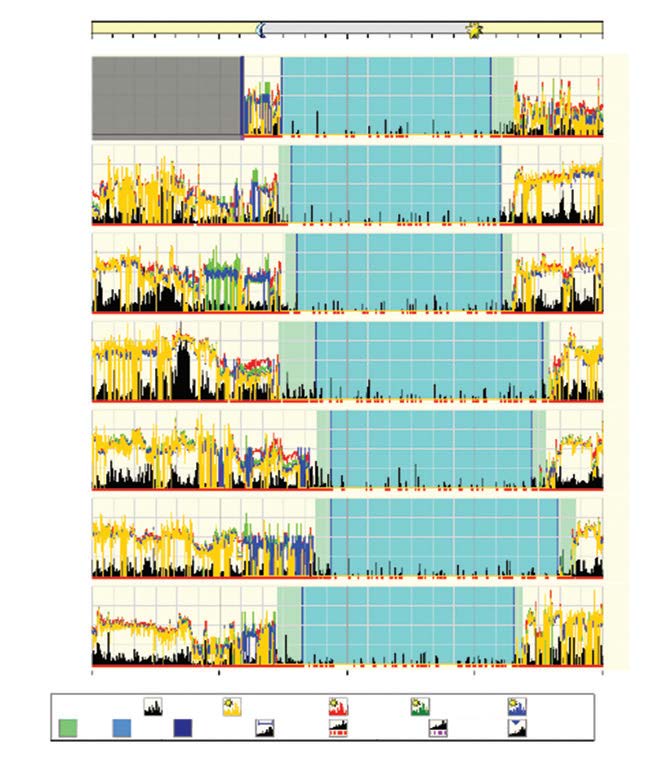
Figure 2. Actigraphy
Delayed sleep phase has been demonstrated in IH but it is not clear if this is coincidental or causal. Regardless, the combination of the two can be very detrimental to social and academic life with consequent mental health implications, as was apparent in our patient.
The etiology of IH is poorly understood. The accumulation of unspecified neurochemical compounds, some genetic markers, immunologic mechanisms, and alteration in circadian molecular mechanisms all have been implicated in the genesis of IH. IH possibly is a cluster of disorders with differing etiologies and natural progressions. Regarding sleep inertia, it has been hypothesized that it occurs when awakening occurs before adenosine, a neuronal metabolite correlated with sleepiness, is fully cleared. The decay of subjective sleepiness after awakening closely parallels the time courses of both extremity cooling and the cortisol awakening response, suggesting possible links of both thermoregulation and the hypothalamic-pituitary-adrenal axis to sleep inertia.
In our case, though AB did not have sleepiness on MSLT or debilitating daytime sleepiness, he had new onset of prolonged sleep time (in contrast with physiological sleep requirements that progressively decrease with age) with extreme sleep inertia that is consistent with IH. AB’s main symptom was sleep drunkenness, particularly on school days and especially when he did not get 11 to 12 hours of sleep.
Management
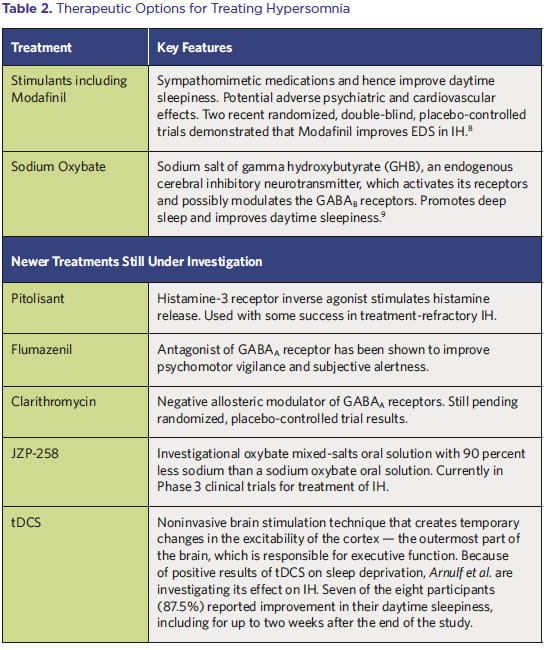
The main goal of treating patients with central disorders of hypersomnolence is to relieve EDS and improve quality of life, and, in the case of IH, to also mitigate sleep inertia.8 Compared to the treatment of narcolepsy, treatment for other hypersomnolence disorders is more challenging and less codified. No medication is currently approved by the United States Food and Drug Administration (FDA) to treat IH; consequently, medications approved for narcolepsy are often used off-label to treat IH.
The therapeutic options for the treatment of hypersomnia are outlined in Table 2.
In AB’s case, the most distressing symptom was sleep inertia. Since stimulants have been reported to be helpful for some patients with IH, AB was prescribed methylphenidate 30 minutes before scheduled wake time and had tangible improvement in sleep inertia. However, this was discontinued because of tachycardia, hypertension, and weight loss. Modafinil did not improve his sleep inertia and was associated with confusion. Sodium oxybate has been shown to improve sleep inertia in patients with IH, possibly through consolidation of overnight sleep. AB has had difficulty obtaining sodium oxybate because of insurance coverage issues.
In addition to medications, management by behavioral therapy can be critical for patients to learn sleep-wake management skills needed to cope with IH. AB reported improvement in quality of life with behavioral therapy, particularly management of delayed sleep phase that was induced by waking up later on weekends. This phase delay was associated with later sleep onset and greater sleep inertia on school days. A common aspect of the difficulties faced by patients with sleep disorders is the mental distress secondary to the lack of public awareness of
sleep disorders. Sleep psychologists and therapists play a critical role in identifying mental health difficulties that may be secondary to a sleep disorder or may be associated with a sleep disorder and help patients cope with these difficulties. Overall, the importance of these associated issues cannot be understated in the management of patients with sleep-related illnesses.
Conclusion and Summary
AB’s case is unique in that he does not fit in a well-defined “box.” His case illustrates the continuum of sleep disorders and the difficulty many patients encounter in obtaining a diagnosis. A diagnosis of IH should only be made after carefully ruling out other causes of EDS. IH causes daytime sleepiness and sleep inertia that can have a tremendous impact on quality of life, especially when associated with delayed sleep phase. Pharmacologic treatment combined with nonpharmacologic intervention offers the best chance of improving quality of life in patients with IH. Although recent work has provided clinicians with some direction in terms of pharmacologic and nonpharmacologic strategies, much work remains to be done.
References
1 Sehgal A, Mignot E. Genetics of Sleep and Sleep Disorders. Cell. 2011; 146(2): 194–207.
2 Medicine AAOS. International Classification of Sleep Disorders — Third Edition (ICSD-3). Darien; 2014.
3 Arnulf I, Groos E, Dodet P. Kleine-Levin Syndrome: A Neuropsychiatric Disorder. Rev Neurol (Paris). 2018; 174(4): 216–227.
4 Roth B. Narcolepsy and Hypersomnia: Review and Classification of 642 Personally Observed Cases. Schweiz Arch Neurol Neurochir Psychiatr. 1976; 119(1): 31–41.
5 Smith-Coggins R, Howard SK, Mac DT, et al. Improving Alertness and Performance in Emergency Department Physicians and Nurses: The Use of Planned Naps. Ann Emerg Med. 2006; 48(5): 596–604.
6 Horne J, Moseley R. Sudden Early-morning Awakening Impairs Immediate Tactical Planning in a Changing “Emergency” Scenario. J Sleep Res. 2011; 20(2): 275–278.
7 Roth B, Nevsímalová S, Ságová V, Paroubková D, Horáková A. Neurological, Psychological and Polygraphic Findings in Sleep Drunkenness. Schweiz Arch Neurol Neurochir Psychiatr. 1981; 129(2): 209–222.
8 Billiard M, Sonka K. Idiopathic Hypersomnia. Sleep Med Rev. 2016; 29: 3–33.
9 Leu-Semenescu S, Louis P, Arnulf I. Benefits and Risk of Sodium Oxybate in Idiopathic Hypersomnia Versus Narcolepsy Type 1: A Chart Review. Sleep Med. 2016; 17: 38–44.
About the Pediatric Sleep Evaluation Center
The Pediatric Sleep Evaluation Center at UPMC Children’s Hospital of Pittsburgh is a multidisciplinary program dedicated to diagnosing and treating all sleep disorders in children, from birth to 21 years of age.
The Center provides care to children with complex issues including craniofacial abnormalities, Down syndrome, ventilator-dependent children, and Prader–Willi syndrome. The Center has two behavioral psychologists embedded in the program, Ryan Anderson, PhD, and Hannah Ford, PhD, allowing comprehensive care for mask desensitization, insomnia, parasomnias, hypersomnia, sleep disruption due to mental/psychiatric disorders, and circadian rhythm disorders. The American Academy of Sleep Medicine-accredited sleep lab at UPMC Children’s has eight beds with a team of sleep technologists led by Craig Halper, RPSGT. The program conducts approximately 1,700 to 1,800 attended in-laboratory-based sleep studies on patients from infancy to young adulthood. The Center provides training in pediatric sleep medicine to fellows from the Sleep Medicine Fellowship at UPMC, which over the last 10 years has trained more than 20 sleep fellows from varied medical specialties, including adult and pediatric pulmonology, adult and pediatric neurology, family medicine, pediatrics, internal medicine, psychiatry, and otolaryngology.
The Pediatric Sleep Evaluation Center works closely with other specialties, including pediatric otolaryngology, plastic surgery, pediatric neurosurgery, and pediatric neurology.
To contact a physician or refer a patient for consultation, please call Melisa Kennedy, administrative coordinator, at 412-692-8736.
Pediatric Sleep Evaluation Center Staff
Hiren Muzumdar, MD - Director
Deepa Burman, MD - Co-Director
Ryan Anderson, PhD
Hannah Ford, PhD
Craig Halper, RPSGT
Frank Boyd, RPSGT
Nancy Popovich, RRT, RPSGT