Password Reset
Forgot your password? Enter the email address you used to create your account to initiate a password reset.
Forgot your password? Enter the email address you used to create your account to initiate a password reset.
4 Minutes
Specialized immune cells that accumulate in the brain in the days and weeks after a stroke promote neural functions in mice, pointing to a potential immunotherapy that may boost recovery after the acute injury is over, University of Pittsburgh School of Medicine neurologists found.
The study, published in the journal Immunity, demonstrated that a population of specialized immune cells, called regulatory T (Treg) cells, serve as tissue repair engineers to promote functional recovery after stroke. Boosting Treg cells using an antibody complex treatment, originally designed as a therapy after transplantation and for diabetes, improved behavioral and cognitive functions for weeks after a stroke in mice compared to those that did not receive the antibody complex.
 “The beauty of this treatment is in its wide therapeutic window,” said senior author Xiaoming Hu, MD, PhD, associate professor in the Department of Neurology at Pitt’s School of Medicine. “With most strokes, you have four and a half hours or less when you can give medication called tPA to reopen a blocked blood vessel and expect to rescue neurons. We’re excited to identify a mechanism that may promote brain recovery by targeting non-neuronal cells well after this window closes.”
“The beauty of this treatment is in its wide therapeutic window,” said senior author Xiaoming Hu, MD, PhD, associate professor in the Department of Neurology at Pitt’s School of Medicine. “With most strokes, you have four and a half hours or less when you can give medication called tPA to reopen a blocked blood vessel and expect to rescue neurons. We’re excited to identify a mechanism that may promote brain recovery by targeting non-neuronal cells well after this window closes.”
Previous research in stroke has been focused on developing new drugs to reduce neuronal death. And whereas these acute stroke treatments quickly lose effectiveness after neurons die, Treg cells remain active for weeks after the injury.
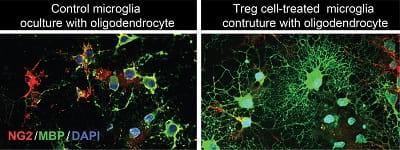 True to their name, Treg cells are immune cells that regulate the immune response, including curtailing excessive inflammation that could harm more than help. Hu and her colleagues observed that the levels of Treg cells infiltrating the brain began to increase about a week after a stroke and continued increasing up to five weeks later. So, they did multiple tests in mice after they’d had strokes, paying particular attention to the brain’s white matter — which is the brain tissue through which neurons pass messages, turning thoughts into actions, like lifting food to your mouth or saying the name of an object you’re looking at.
True to their name, Treg cells are immune cells that regulate the immune response, including curtailing excessive inflammation that could harm more than help. Hu and her colleagues observed that the levels of Treg cells infiltrating the brain began to increase about a week after a stroke and continued increasing up to five weeks later. So, they did multiple tests in mice after they’d had strokes, paying particular attention to the brain’s white matter — which is the brain tissue through which neurons pass messages, turning thoughts into actions, like lifting food to your mouth or saying the name of an object you’re looking at.
Mice that were genetically unable to produce Treg cells fared worse than mice with a robust Treg cell response. Interestingly, it was only in the latter phases of stroke recovery that the Treg cell-depleted mice suffered impairments in white matter integrity and behavioral performance compared to mice with a normal Treg cell response.
Additionally, when normal mice were given an antibody complex called “IL-2:IL-2Ab” to boost their Treg cell levels after a stroke, their white matter integrity improved even more and neurological functions were rescued over the long term. The mice with more Treg cells had an easier time moving and had better memories, allowing them to navigate mazes faster after a stroke than their non-treated counterparts.
“This strongly suggests that, rather than working to preserve white matter structure and function immediately after a stroke, Treg cells influence long-term white matter repair and regeneration,” said Hu, also a member of the Pittsburgh Institute of Brain Disorders and Recovery and a U.S. Department of Veterans Affairs (VA) investigator. “Our findings pave the way for a therapeutic approach to stroke and other neurological disorders that involve excessive brain inflammation and damage to the white matter. Treg cells appear to hold neurorestorative potential for stroke recovery.”
Hu stressed that there are still many hurdles to cross before Treg cells could be used in humans for stroke recovery. Namely, research is needed to determine the best way to boost the number of Treg cells in stroke victims. This could be done by improving IL-2:IL-2Ab so that it better stimulates production of Treg cells with fewer side effects, or a personalized therapy could be developed where some cells are taken from an established Treg cell bank and used to grow custom Treg cells in the lab, which could then be infused back into the patient.
Additional authors on this research are Ligen Shi, MD, PhD, Zeyu Sun, MD, Wei Su, MD, Di Xie, MD, Qingxiu Zhang, MD, Xuejiao Dai, MD, PhD, Kartik Iyer, BS, T. Kevin Hitchens, PhD, Lesley M. Foley, BS, Sicheng Li, PhD, Donna B. Stolz, PhD, Kong Chen, PhD, Ying Ding, PhD, and Angus W. Thomson, PhD, all of Pitt; Fei Xu, B.S., and Jun Chen, MD, both of Pitt and the VA Pittsburgh Health Care System, and Rehana K. Leak, PhD, of Pitt and Duquesne University.
This research was supported by the National Institute of Neurological Disorders and Stroke grant NS094573 and VA grant I01 BX003651.