Password Reset
Forgot your password? Enter the email address you used to create your account to initiate a password reset.
Forgot your password? Enter the email address you used to create your account to initiate a password reset.
6 Minutes
This case study was conducted by Hussain Mahmud, MD, and Nami Safai Haeri, MD, both from the Division of Endocrinology and Metabolism.
Acute hemorrhage of the adrenal gland, without prior history of trauma or anticoagulation, is known as spontaneous adrenal hemorrhage (SAH). SAH usually presents with nonspecific symptoms such as fatigue, dizziness, nausea, abdominal pain, and anorexia. SAH can easily be mistaken for other conditions that present with shock or acute abdominal pain. While 90% of SAH cases are unilateral, and statistically more common in the right adrenal gland, there are rare occurrences of bilateral SAH.1 Two cases of bilateral SAH that occurred during the third trimester of pregnancy are discussed in this report.
A 28-year-old female with a history of irritable bowel syndrome (IBS) and asthma presented to a UPMC emergency room with nausea, vomiting, and right sided abdominal pain during the 35th week of her first pregnancy. An abdominal MRI revealed a 4.7 x 2.8 cm right adrenal hemorrhage (see Image 1).
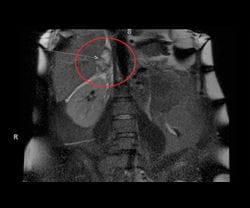
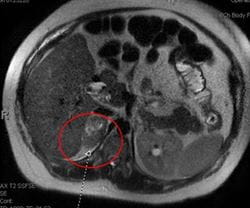
Image 1: MRI without contrast remarkable for a 4.7 x 2.8 cm indeterminate right adrenal lesion with associated perinephric fluid. This finding is concerning for adrenal hemorrhage.
Her vital signs were stable on presentation. Laboratory results revealed sodium 134 mEq/L, potassium 3.4 mEq/L, hematocrit 33.4%, and a random cortisol level of 26 mcg/dL. She was admitted to the hospital for observation. Over the next 24 hours her symptoms improved and she remained clinically stable. She was discharged and advised to follow up with an endocrinologist and her obstetrician within four weeks.
She returned to the emergency room four days after discharge with similar abdominal pain, this time occurring on the left side. A second MRI, however, did not show adrenal bleeding. Due to the patient’s blood pressure (BP) of 90/70 mmHg, hydrocortisone IV 50 mg was administered every eight hours. Thirty-six hours following the inpatient admission, the patient became tachycardic and tachypneic.
A CT angiogram (CTa) ruled out a pulmonary embolus (PE), but revealed an interval development of a 5.3 x 3 cm left adrenal hemorrhage (see Image 2).
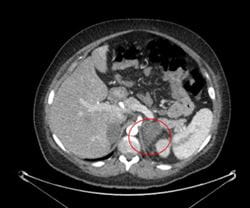
Image 2: CT angiogram remarkable for interval development of a 5.3 x 3 cm left adrenal mass with subtle infiltration of adjacent fat, consistent with left adrenal hemorrhage.
The patient had normal sodium and potassium levels with no findings indicative of Primary Adrenal Insufficiency (PAI) and was discharged to home on a physiologic dose of hydrocortisone.
A 30-year-old female with a history of hypertension, cerebral aneurysm, and polycystic kidney disease presented to a UPMC emergency room with nausea and left side abdominal pain during the 31st week of her second pregnancy. An abdominal CT scan showed a 2.3 x 3.1 cm left adrenal hemorrhage (see Image 3).
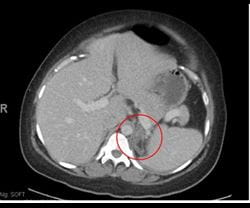
Image 3: CT without contrast remarkable for enlarged left adrenal gland with adjacent fat stranding. Findings are concerning for left adrenal hemorrhage.
At the time of the patient’s hospital admission, her BP was 85/50 mmHg. Therapy with IV hydrocortisone was initiated at an initial dose of 50 mg, followed by 25 mg administered every eight hours. Her BP subsequently improved (112/68 mmHg) within 24 hours. Three days after the initial presentation, the patient developed new right flank pain. The MRI was remarkable for features indicative of adrenal hyperplasia but did not confirm presence of hemorrhage.
The following day, the patient developed hypoxia and tachycardia. A follow-up CTa was remarkable for the development of a new right adrenal hemorrhage (see Image 4).
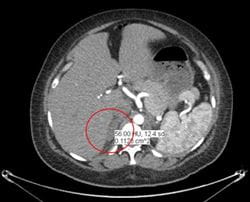
Image 4: CT Angiogram remarkable for increased soft tissue bulkiness and partial obliteration of fat planes within the region of the right adrenal gland with Hounsfield units measuring 56 suspicious for new right adrenal hemorrhage.
She did not have hyperkalemia or other findings indicative of PAI and was discharged to home on 30 mg total daily dose of hydrocortisone.
SAH is extremely rare with an estimated incidence of 0.03% to 1.8% based on autopsy reports in patients with complaints of abdominal pain. Spontaneous adrenal hemorrhage in pregnancy can lead to adrenal crisis, often associated with a high rate of fatality in both the mother and fetus. Some pregnancy-related causes for adrenal hemorrhage are spontaneous abortion, ovarian cyst torsion, preeclampsia, antepartum or postpartum hemorrhage, and coagulation disorders, such as antiphospholipid syndrome.1
During pregnancy, there is adrenal cortex hyperplasia and hypertrophy secondary to physiological elevation of the adrenocorticotropic hormone (ACTH) that may predispose a patient to adrenal venous congestion. Increased catecholamine release also constricts the venules and enhances platelet aggregation. The synergistic effect of these two mechanisms increases pressure within the venous sinusoids, especially as the pregnancy progresses, and serves as the likely etiology for increased risk of SAH during pregnancy.2
Common manifestations of SAH include those similar to what is described with an adrenal crisis, including abdominal pain, fever, fatigue, dizziness, arthralgias, myalgias, anorexia, nausea, vomiting, and hypotension. Laboratory findings may reveal anemia, prolonged International Normalized Ratio (INR), and leukocytosis. If SAH is unilateral, it usually does not cause adrenal insufficiency. In patients with bilateral SAH, the presentation is consistent with an acute adrenal crisis. Features of PAI such as hyponatremia and hyperkalemia and a non-anion gap metabolic acidosis can be anticipated when over 90% of adrenal cortices are affected.2
Ultrasonography is usually the initial imaging modality ordered during pregnancy if SAH is suspected, though sonographic features of adrenal hemorrhage are very nonspecific. CT scans have higher sensitivity to detect hemorrhage in the acute phase. MRI has high sensitivity and specificity for SAH although it is a more expensive detection method as compared to other testing modalities. Both MRI or CT scans can also be used to evaluate potential underlying etiology for hemorrhage, such as pheochromocytoma or presence of an adrenal mass.3
For management, patients need to be closely monitored by an endocrinologist and a maternal fetal medicine specialist. Patients are usually conservatively managed with oral glucocorticoid therapy, with or without mineralocorticoid replacement. If indicated, however, intravenous fluid resuscitation and pain control should be administered. Additional management methods include correction of electrolytes, close continued monitoring of hemoglobin, correction of coagulopathy as needed, and careful observation for features of adrenal crisis, such as circulatory collapse, generalized weakness, hypoglycemia, and altered mental status. In pregnant patients, frequent fetal monitoring and obstetric exams are also necessary.2,4
The possibility of the development of bilateral SAH should always be considered among patients presenting with unilateral SAH. If left unrecognized, SAH is associated with poor outcomes and complications for both mother and fetus.
Please refer to the Update in Endocrinology Fall 2021 CME course on UPMCPhysicianResources.com for a full list of references featured in this article.